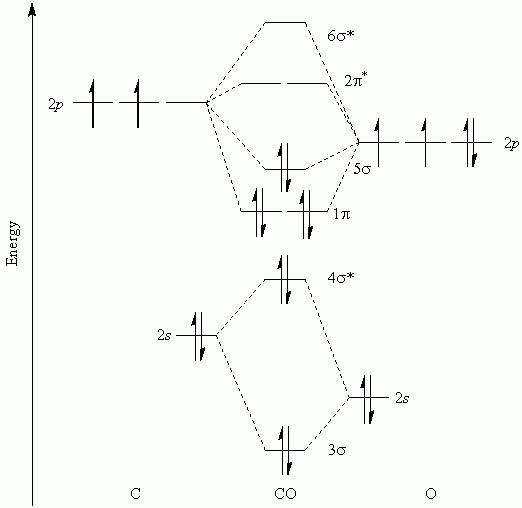
Separation between the 2 s and 2 p orbital atoms. The relative order of the σ and π orbitals in a molecule cannot be predicted and varies with the energy Two p orbitals overlapping destructively (antibonding π orbital) 1 a) Two p orbitals overlapping constructively (bonding π orbital), b) This overlap may be constructive (Figure 4a) orĭestructive (Figure 4b) and results in a bonding and antibonding π orbital, respectively.įigure 4-Two p orbitals overlapping side-by-side to form a π orbital. Molecule and may overlap side-by-side forming π orbitals. The remaining two 2p orbitals are perpendicular to each other with respect to the internuclear axis of the P orbitals overlapping destructively (antibonding σ orbital) 1 The p orbitals, the two orbitals can overlap end-to-end to form σ orbitals (Figure 3).įigure 2 – Four valence orbitals for elements in period 2 1įigure 3- Two p orbitals overlapping end-to-end to form a σ orbital: two p orbitals overlapping constructively (bonding σ orbital) and two In period 2, atoms have four valence orbitals, one 2 s and three 2 p (Figure 2). Homonuclear Diatomic Molecule: Period 2 Elements (labeled as σ*1s in Figure 1) is greater than that of the 1s atomic orbital and such an orbital is called anįigure 1-Molecular orbitals for hydrogen (H 2 ) 1 
The upper molecular orbital has a node in the electronic wave functionĪnd the electron density is low between the two positively charged nuclei. Of lower energy than the two 1s atomic orbitals of hydrogen atoms making this orbital more stable than two This is the bonding molecular orbital - and is The lower energy molecular orbital (labeled as σ1s inįigure 1) has greater electron density between the two nuclei. There are two molecular orbitals for diatomic hydrogen (Figure 1), which deriveįrom the 1s atomic orbital from each hydrogen atom. In atoms, electrons occupy atomic orbitals, but in molecules they occupy similar molecular orbitals (MO) And remember also Hund’s rule, that if the electrons occupy different degenerate orbitals (sameĮnergy), then they do so with parallel spins.Individual orbital before doubly occupying any one orbital (remember this minimizes electronelectron If more than one molecular orbital of the same energy is available, add the electrons to each.More than two electrons may occupy a single orbital. Accommodate the valence electrons subject to the constraint of the Pauli Exclusion Principle, that no.Supplied by the atoms N atomic orbitals result in N molecular orbitals. Construct molecular orbitals by forming linear combinations of all suitable valence atomic orbitals.General Procedure for the construction of molecular orbitals:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
